| |
Заведующий – Л.П. Овчинников
The protein composition of mammalian mRNPs (informosomes) has been determined (Fig. 1).
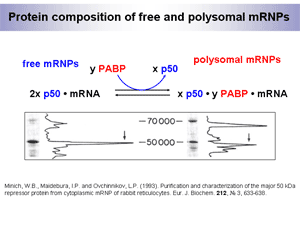
It was shown that free non-translated mRNPs contain one 50 kDa major protein (p50), while polysomal mRNPs have two major proteins, p50 and 70 kDa poly(A)-binding protein (PABP). The amount of p50 in polysomal mRNPs is twice as low as in free mRNPs. Thus,
i) p50 appears the universal major mRNP protein, and
ii) mRNA transition from its non-translated to translated state is accompanied by attachment of PABP and dissociation of half of p50.
Fig. 2. The amino acid sequence of the universal major mRNP protein p50 has been determined.

It appeared that
i) p50 is identical to Y-box binding transcription factor YB-1 as to its amino acid sequence;
ii) p50/YB-1 consists of three domains. Its central domain is extremely evolutionarily conserved and widely distributed from bacteria to human beings. At present, this domain is termed the cold chock domain (CSD).
iii) both CSD and the C-terminal domain display affinity for nucleic acids. Besides, in the absence of mRNA, the C-domain is capable of multimerization.
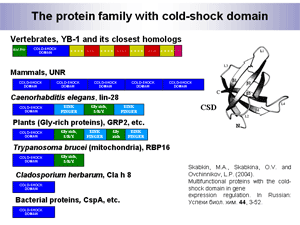
iv) YB-1 is a member of the large cold-shock domain protein family (Fig. 3).
It was proposed and experimentally supported that when interacting with mRNA, YB-1 alone, without other mRNP proteins, gives RNA-protein complexes displaying all unique physico-chemical properties of natural mRNPs. Thus, YB-1-mRNA complexes may serve as the simplest model for studies of the structural organization of natural mRNPs. It was shown that depending on the YB-1/mRNA ratio, YB-1 changes its quaternary structure, mode of its interaction with mRNA, and the overall mRNP packaging (Fig.4).
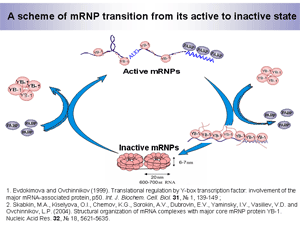
At comparatively low YB-1/mRNA ratios typical of polysomal translated mRNPs, YB-1 binds to mRNA as a monomer through its both RNA-binding domains (CSD and C-domain) and form an unfolded translationally active mRNP structure. Upon mRNA saturation with YB-1, which is characteristic of free non-translated mRNPs, YB-1 interacts with mRNA predominantly through the cold shock domain. The displaced C-domains undergo multimerization. Most probably, mRNA is located on the surface of these YB-1 multimers, since it is extremely sensitive to endoribonucleases. One YB-1 multimer (~600 kDa) packs an mRNA segment of 600-700 nucleotide residues. More extended mRNA molecules and YB-1 form polyparticles. In such complexes, mRNA is translationally inactive and stabilized both in lysates and in vivo, probably due to inaccessibility of mRNA termini for exoribonucleases that usually trigger mRNA degradation.
|
|
